 
Our pages contain various quotes with which our editorial team does not always agree. See also: Sulfoxide, Galantamine, Electrode potentials, Fuming sulfuric acid, Allotropic Principles of Chemistry Molecular Approach. LIMITED TIME OFFER: GET 20 OFF GRADE+ YEARLY SUBSCRIPTION. The liberated tag could then be resolved and detected as subpicomole concentrations using capillary gas chromatography EC-GC. Electron capture dissociation (ECD) is a promising method for de novo sequencing proteins and peptides and for locating the positions of labile posttranslational modifications and binding sites of noncovalently bound species. Get the detailed answer: Determine the identity of the daughter nuclide from the electron capture by 81 37 Rb. Once the halo aromatic analyte was attached to the bead it could be selectively detached into solution upon photolysis with UV light. In positron emission or in, Z decreases. There are many other rarer modes of decay.Ī particular nucleus may decay by more than one mode.ġ) A radioactive process in which the atomic number, Z, of the radioactive nuclide changes but the mass number, A, does not. Z backbone (as drawn), nuclei have too few neutrons and either undergo or positron emission decay. (EC) - form of radioactive decay in which the atomic nucleus absorbs a K or L shell electron, converting a proton into a neutron.Įlectron cloud - region of negative charge surrounding the atomic nucleus that has a high probability of containing electrons. The decay of an unstable nucleus by capturing, or picking up, an electron from an inner orbital of an atomĮlectron configuration the particular distribution of electrons among available subshells. An electron interacts with an up quark in the nucleus via a W boson to create a down quark and electron neutrino.ĭetector - One of the most sensitive gas chromatographic detector for halogen-containing compounds like chlorofluorocarbons. The leading-order Feynman diagrams for electron capture decay. Electron capture is a process in which the proton-rich nucleus of an electrically neutral atom absorbs an inner atomic electron, usually from the K or L. The decay scheme for electron capture is. This process reduces the atomic number by 1 and emits gamma radiation and a neutrino. The loss of an inner shell electron leaves a. 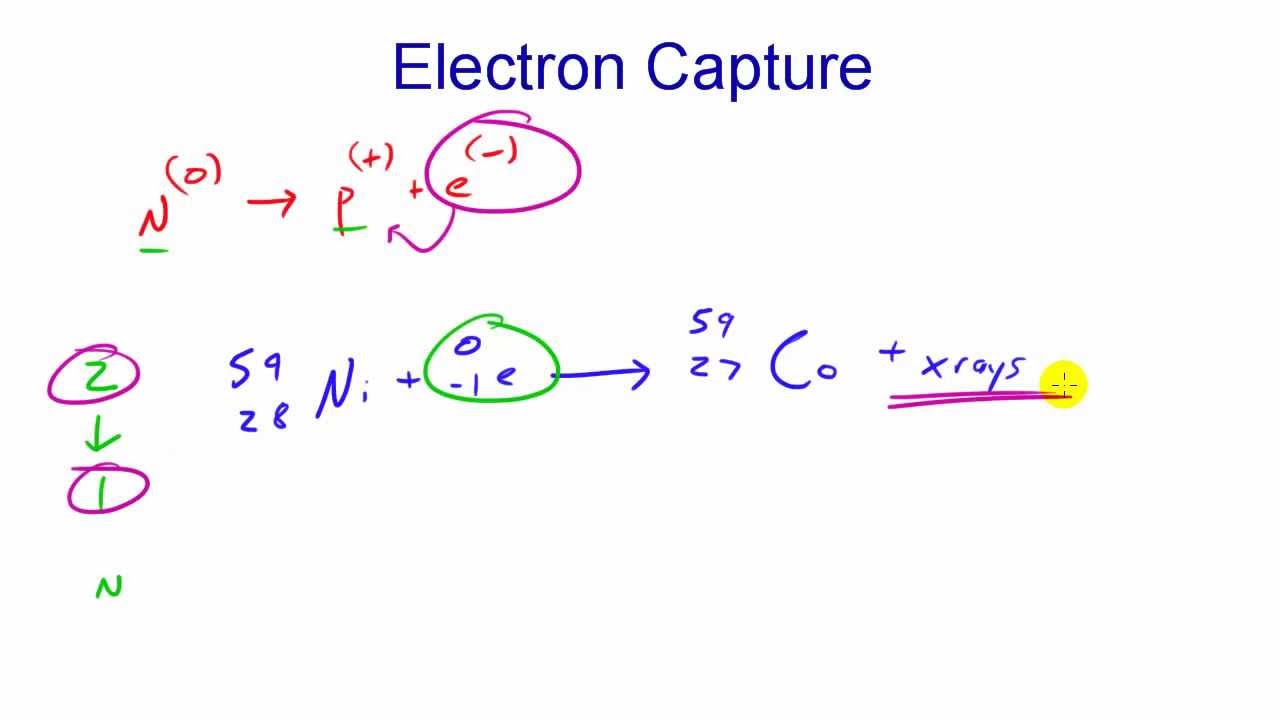
The electron converts a proton to a neutron in the nucleus.Įlectron capture - Electron capture is a type of radioactive decay where the nucleus of an atom absorbs a K or L shell electron and converts a proton into a neutron. Electron capture occurs when an inner shell electron combines with a proton and is converted into a neutron. Electron capture: capture of an electron by an unstable nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
